Biological and Health Sciences
Small molecules, great advances
Argentine and German scientists found how a protein regulates the growth of some type of tumours. This finding would be used to develop localized therapies.
In 2007, the team coordinated by Eduardo Arzt, CONICET senior researcher and professor at the Faculty of Exact and Natural Sciences of the University of Buenos Aires, identified for the first time the genetic code for RSUME protein and discovered that one of the roles of this molecule is to modify proteins to assign new roles, for instance the ability to mediate the cell’s response to external factors such as lack of oxygen, etc.
Currently, Arzt leads the Biomedicine Research Institute of Buenos Aires (IBioBA, CONICET-Partner Institute of the Max Planck Society). Together with his team, the researcher has been studying how RSUME modulates the activity of the protein encoded in the VHL gene, which is associated with the von Hippel-Lindau syndrome (VHL), a disease characterized by the formation of highly vascularised tumours in different organs such as the kidney, cerebellum and adrenal glands.
A recent study published in Oncogene magazine showed how RSUME interacts with VHL and regulates the adaptation of tumour cells and their proliferation.
“RSUME acts in various processes”, Arzt explains and adds “one of them is to regulate the cell’s response to the lack of oxygen – hypoxia -, something that happens frequently in tumours”. As these cells proliferate, they need more nutrients and oxygen that come from blood. Nevertheless, the normal speed of the production of new vessels is not enough to satisfy the demand of the tumour, which ends up surrounded by hypoxic niches. VHL patients, however, have highly vascularised tumours, that is to say, that these cells have this adaptation mechanism activated.
For this reason, one of the questions that arose was ‘how do these tumour cells survive in these environments with low oxygen concentrations?’ The answer was provided after the analysis of the samples that belonged to von Hippel-Lindau syndrome patients and with the help of RSUME and VHL.
It was not new that the molecules that regulate the cell’s response to the reduction in the oxygen levels are the Hypoxia Inducible Factors (HIFs). These ones activate protein production to promote the adaptation of the cell to environments with low pressure oxygen. Without the HIFs, this process is not possible. “It was known that the sign for the HIF to degrade was through the ‘targeting’ with a small molecule called ubiquitin”, Arzt explains. The last stage of this process is regulated, in the case of the HIF, by VHL, the same molecule whose gene is mutated in the von Hippel-Lindau syndrome patients.
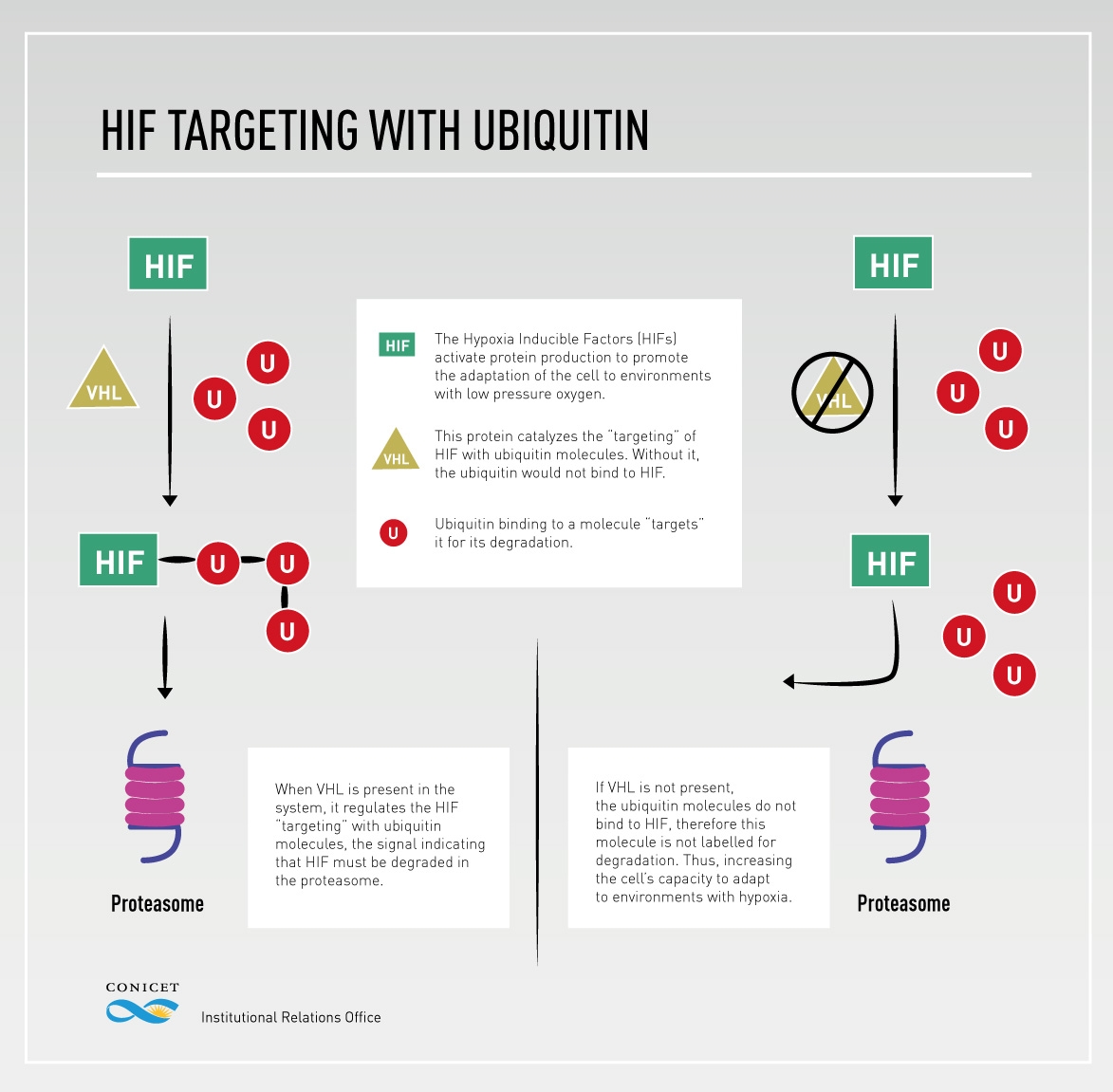
(Click on the image to enlarge)
The results of the study demonstrated that RSUME inhibits VHL, thus preventing HIF degradation, what allows cells to adapt faster to the lack of oxygen; and avoiding tumour proliferation.
One step beyond
For the research team, the study has two main components: first, it describes the RSUME/VHL mechanism of action at a molecular level, but apart from that it shows how the cells with VHL mutations need RSUME to proliferate.
Patients suffering from this disease have hemangioblastomas, which are central nervous system tumours; pheochromocytomas, the ones that are in the adrenal glands; and renal cell carcinoma, and others.
“After analyzing the RSUME expression patterns, we found that the tissues with higher levels were those ones affected by the von Hippel-Lindau syndrome: adrenal gland, cerebellum and kidney”, Lucas Tedesco, CONICET doctoral fellow at the IBioBA and one the authors of the research, comments.
This information led us to the development of a hypothesis: it was known that the RSUME affected HIF, and that its expression patterns were similar to the one of the von Hippel-Lindau syndrome, so it was probably that the RSUME acted on the VHL to regulate HIF and promoted the adaptation of the tumoral cells and its proliferation.
In order to test the hypothesis, the researchers implanted VHL tumour cells in two groups of mice: one of them expressed RSUME and the other did not. “We noticed that those mice in which the RSUME was present developed a greater number of tumours characteristic of VHL syndrome, while those ones without RSUME had smaller and less vascularised tumours”, Tedesco explains. Besides, the scientists noticed that in the case of the last group the proliferation of malignant cells was reduced to 50 per cent approximately.

(Click on the image to enlarge)
In spite of the promising results, the researchers state that they still need more studies to develop a therapy based on this knowledge. “Any treatment that includes RSUME should be thought to be used locally and selectively because this molecule is present in various tissues”, Juan José Bonfiglio, PhD in Biology and author of the study, adds.
In future researches, the scientists will seek to further characterize the effects of RSUME on VHL and other proteins. Finally, Arzt comments “for us, one of the most motivating aspects of this study is that between 60 and 70 percent of the ‘lab bench’ work was done at the IBioBA, what makes our study real”.
Cooperative work
One of the authors of the research is Marta Barontini, PhD in Medicine and CONICET Senior researcher. She works at the Laboratorio de Estudios del Sistema Nervioso Autónomo del Centro de Investigaciones Endocrinológicas (CEDIE, CONICET-FEI) and at the “Dr. Ricardo Gutiérrez” Children’s Hospital. The samples used during the research belonged to VHL syndrome patients.
- By Ana Belluscio.
- About the research
- L. Gerez. IBioBA. FCEyN-UBA.
- L. Tedesco. Doctoral fellow. IBioBA.
- JJ Bonfiglio. IBioBA.
- M. Fuertes. IBioBA.
- M. Barontini. Senior researcher (r). CEDIE.
- S. Silberstein. Independent researcher. IBioBA. FCEyN-UBA.
- Y. Wu. Max Planck Institute of Psychiatry, Munich. Germany.
- U. Renner. Max Planck Institute of Psychiatry, Munich. Germany.
- M. Páez-Pereda. Max Planck Institute of Psychiatry, Munich. Germany.
- F. Holsboer. Max Planck Institute of Psychiatry, Munich. Germany.
- GK Stalla. Max Planck Institute of Psychiatry, Munich. Germany.
- E. Arzt. Senior researcher. IBioBA. FCEyN-UBA.
